Q: I've been told that drinking water should be alkaline, but BEV values allow for slight acidity... how do you explain that?
A: There are a number of companies currently manufacturing water treatment devices which alter water properties creating acidic and alkaline pH values. We discuss the merits of these devices on another page. To answer the question above, a little understanding of pH (acid/base) chemistry is required. We will do our best to avoid being overly technical:
pH is a relative measure of the degree of acidity or alkalinity of a solution, in our case water. Some substances, such as hydrogen ions, lower the pH of a solution. They are considered acids. Other substances, such as hydroxide ions, raise the pH of a solution. Those are called bases. The pH scale ranges from 0 - 14, with 7 being considered the mid or neutral point. pH below 7 is considered acidic, and above 7 is considered alkaline.
As an example, the pH of apple juice is typically 2.9 - 3.3, and the acid in our stomachs is typically between 1 and 3. Milk of magnesia has a pH ~10.5, household bleach is ~12.6, and ammonia is typically ~11.5.
 |
|
Basic shape of water molecule. |
|
There are many different acids and bases that affect pH, but since we are talking about water we will limit our discussion to hydrogen ions (H+) and hydroxide ions (OH-). It should be apparent these are the components of a water molecule, or H2O. When water has more hydrogen ions floating around than it does hydroxide ions, it is acidic (pH less than 7). When there are more hydroxide ions than hydrogen ions, it is basic (pH greater than 7).
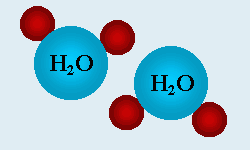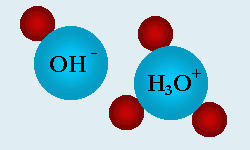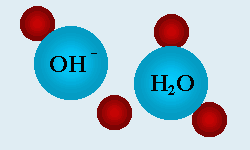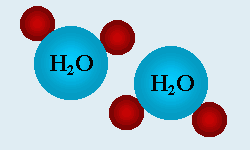 |
If we were to examine pure water at the molecular level, we would see the water molecules do not stay bound together as H2O, but are instead constantly separating and recombining with each other. A single H2O will break into an H+ and OH-, create intermediary forms of H3O+ and OH-, and then get back together again. This happens constantly to the trillion upon trillions of water molecules. In extremely pure water, the number of H+ and OH- would always be equal, and the pH would be neutral at 7. There are no other substances present to influence the pH in either the acidic or alkaline direction.
So why do the BEV values allow for the pH to be acidic?
This is because Prof. Vincent found the most important quality of biocompatible water is its purity, and the purity which makes the water neutral also makes it extremely sensitive to the addition of other substances. Chemically speaking, the extreme purity of BEV water means it has no buffering capacity. This means the addition of very small amounts of an acid or base will cause large effects on pH.
As an example of buffering capacity, consider sitting alone in a very quiet room with music playing softly on the stereo. You do not need a lot of volume to be able to hear the music. Small increases in volume are easily noticed. Now consider bringing the noise of downtown Manhattan into your quiet room—you will have to turn the volume up to extremely high levels to hear the music.
The empty quiet room is like the glass of extremely pure BEV water. If only a tiny amount of acid is added, you will be able to notice the difference in pH. The noisy room is like a glass of tap water before purification, full of all kinds of contaminants. Adding just a little acid or base will have little effect on pH, just as adding more volume to the stereo will go unnoticed in the noisy room.
Untreated tap water typically contains chemicals that act as "buffers." When a little more H+ is added, some of the buffers attach to the H+ and combine with it. Then, even though we've added more acid, the pH doesn't change because the amount of "free" H+ is still relatively the same as the amount of free OH-. Because the purification process in the PWS™ BEV Series systems removes the buffering chemicals, addition of a very, very small amount of H+ will easily upset the balance between the free H+ and free OH-.
Due to the extreme purity of BEV quality water, it becomes a super-solvent, quickly dissolving carbon dioxide from the air. The carbon dioxide molecules combine with some of the free OH- ions in the water. This leaves more free H+ ions, which is how we define an acidic solution. This is why the pH of BEV quality water can be on the acid side of neutral.
Remembering the pH scale measures the relative concentration, it then becomes obvious we are describing an extremely weak acid solution. Having an acidic reading in BEV water should not be cause for concern because of this extremely weak acidity. The slightest speck of a buffer such as baking soda (a base) would quickly neutralize the slight acidity in the BEV water. It requires only 10mg/l of baking soda to move the pH of pure BEV water 3 full pH points. Certainly, we don't recommend adding a spec of baking soda to each tank of water. Once pure water mixes with saliva, the pH values will change instantly.
(The acid pH readings would be very different if we had the same pH reading in untreated tap water containing a large quantity of buffering chemicals—in that case we would be describing a much stronger acid solution, which would require a great deal more base to neutralize.)
Obviously, we don't recommend you go to the trouble of trying to adjust the pH of every glass of water. The instant the extremely pure BEV water touches your tongue it begins to combine with the saliva in your mouth, and just moments later it combines with the stomach contents. Remembering our stomach acidity is typically ~ pH of 2 (considered highly acidic), the extremely pure BEV water will no longer have the unique pH characteristics described above.
(It is important to note that some stomach enzymes, like pepsin which begins the breakdown of protiens, only function in this extremely acidic environment. Further, the extreme acidity in our stomachs kills many bacteria which we might consume with our food.)
The only way drinking the slightly acidic pH BEV quality water could possibly upset the pH balance in the body might be if you were to drink ridiculously large amounts, and didn't eat any food, and kept that up for a long time. Barring that situation, drinking the very weakly acidic pH BEV quality water will have no effect on the pH of your body fluids.
How does this relate to alkaline water?
Alkaline water machines do not purify the water. Most offer some degree of carbon filtration, but this does not remove the minerals (buffering agents) in the water. These minerals, if not present, must be added back to extremely pure water in order to separate the water into acid and alkaline components. This mineral content does not equal the exteme purity Prof. Vincent found so important in biocompatible water. Remember too, just like a weak acid, you can easily create a weak alkaline solution with the addition of very small quantities of base (assuming you begin with extremely pure water).
However, it is vitally important to remember the BEV values do not consider pH alone, but also rH2, which is a linear function of pH. Therefore, if the pH value drifts too far to the alkaline side of the scale, the rH2 value will fall out of range. Prof. Vincent considered biocompatible water to not only be extremely pure with the correct pH reading, but it must also have the correct rH2 reading, providing an indication of the overall vitality of the water. Considering pH alone does not provide enough information to determine whether water is biocompatible or not.

Please see our “Special Notice” for details on an exciting new Pure Water Systems product to increase alkalinity of ultra-pure BEV quality water.